Ascites represents a particularly valuable source of tumour material as it enables minimally invasive and repeatable sampling of metastatic disease, yields substantially higher numbers of viable tumour cells than solid tumour biopsy and is associated with improved PDC establishment success rates. Unlike solid tumour processing, ascites derived cultures require minimal tissue dissociation, reducing processing associated cellular stress and accelerating model establishment whilst preserving tumour and microenvironment associated populations. Importantly, ascites drainage can often be performed serially throughout disease progression, enabling longitudinal sampling from the same patient and supporting investigation of tumour evolution, therapeutic response and resistance over time.
Our approach and expertise
Our group has established a robust and reproducible workflow for the generation of patient derived cellular models from malignant ovarian ascites, with current establishment success rates approaching 75%. This has been achieved through optimisation of patient selection, sample processing and epithelial selective culture conditions specifically designed to maximise tumour cell viability whilst limiting stromal overgrowth.
Samples are preferentially collected from patients with metastatic disease and at least four weeks post chemotherapy, reducing treatment associated cytotoxicity and increasing the likelihood of recovering viable proliferative tumour cells. Ascites samples are processed rapidly following collection using low passage workflows that preserve cellular heterogeneity and minimise culture induced drift.
A key strength of the platform is the use of a serum free epithelial supportive medium formulation optimised in house to selectively support epithelial tumour cell attachment and expansion. Early cultures undergo differential trypsinisation to exploit the differing adhesion kinetics of fibroblastic and epithelial populations, enabling efficient depletion of stromal contaminants whilst preserving tumour enriched epithelial colonies. These conditions support the establishment of highly proliferative epithelial cultures and three dimensional spheroid formation characteristic of ovarian tumour biology.
The images below show the different cell types we have successfully isolated and enriched from a single donor sample.
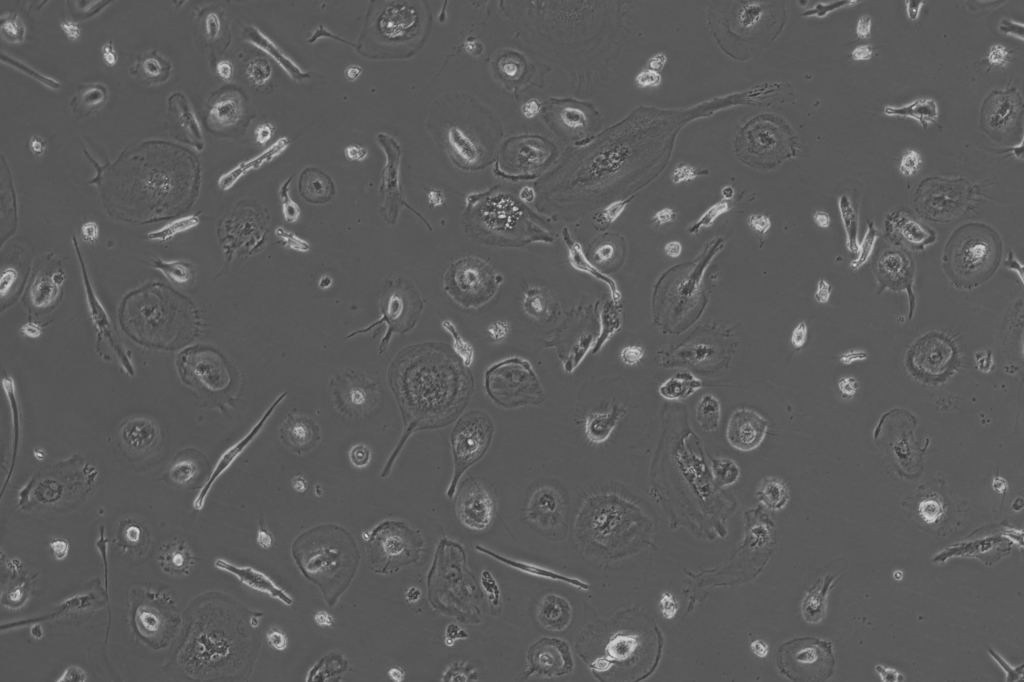
Mesothelial cell enriched population 10x
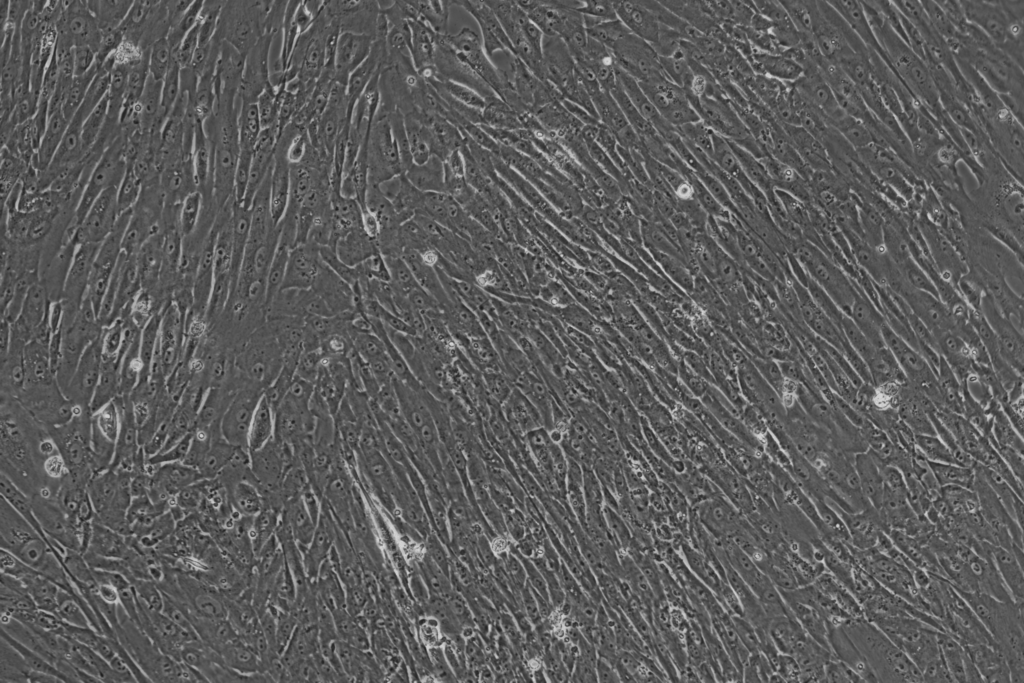
Stromal/fibro enriched population 10x

Epithelial/cancer enriched population 10x
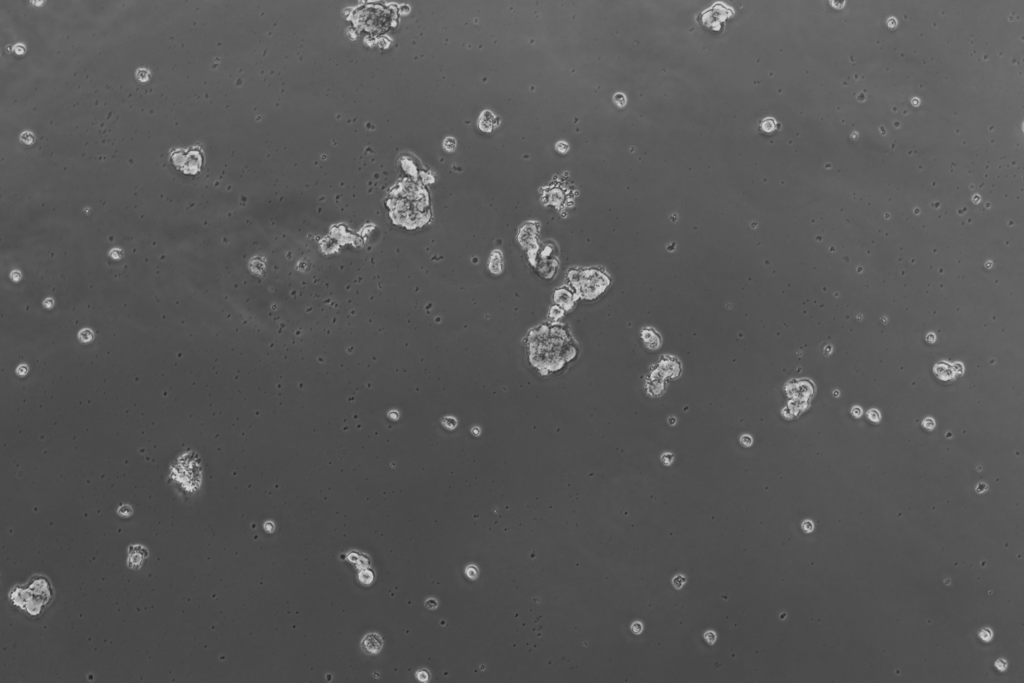
Epithelial/cancer enriched spheroids 10x
In addition to tumour cell isolation, we routinely process and preserve matched acellular ascites fractions following sterilisation and filtration. These patient matched supplements can be reintroduced where appropriate to support physiologically relevant culture conditions, particularly for the enrichment and maintenance of stromal populations and tumour microenvironment studies. We also isolate and preserve mesothelial cell populations from ascites drains, recognising their important role in ovarian cancer dissemination, metastatic niche formation and therapeutic response within the peritoneal cavity.
Together, this integrated workflow enables the generation of low passage clinically relevant ovarian PDC models whilst preserving key components of the native ascites microenvironment for downstream functional, translational and co culture applications.
To find out more, get in touch with our team.
